Beckman Coulter AU 640 Chemistry Analyzer
12000$
Model Number AU640 Event Date 07/25/2011 Event Type Malfunction Event Description Printout of hdl (high-density lipoproteins) results for the day showed low result, defined as <20, from 1600-1730. Consulted manufacturer; results from before and after the problem period were rerun. Results previous to 1600 and after 1730 all reproduced. The low results from 1600 to 1730 had corrected reports issued and physicians notified verbally and documented in lis. Beckman coulter started a cf type 1 fda investigation which is on going. Approximately 25 patients had corrected results issued. Brand Name OLYMPUS AU640 Type of Device LAB CHEMISTRY ANALYZER Manufacturer (Section D) BECKMAN COULTER 3131 w. royal lane irving TX 75063 MDR Report Key 2195287 Report Number 2195287 Device Sequence Number 1 Product Code JJE Report Source User Facility Type of Report Initial Report Date 08/03/2011 1 Device Was Involved in the Event 0 PatientS WERE Involved in the Event: Date FDA Received 08/03/2011 Is This An Adverse Event Report? No Is This A Product Problem Report? Yes Device Operator Invalid Data Device MODEL Number AU640 Was Device Available For Evaluation? Yes Is The Reporter A Health Professional? No Answer Provided Was the Report Sent to FDA? Yes Date Report Sent to FDA 08/03/2011 Event Location Hospital
related Products

spectrophotometer 720 icp varian
10500 USD
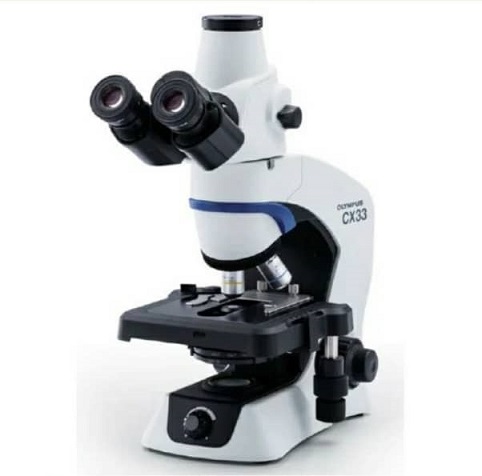
Olympus cx33
650 EUR

intra gastric balloon kit
500 EUR

intra gastric balloon kit
980 EUR

Hydrogen peroxide cassette
56 EUR

CoaDATA 504
580 USD
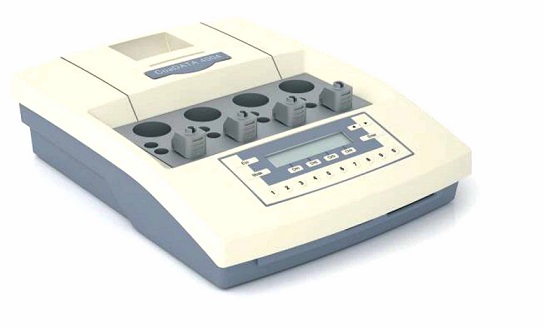
CoaDATA 4004 and 4-channel coagulation analyser
520 USD

Laboratory heater Industrial Model N-21 2A 250v
510$

Laboratory heater Industrial Model N-21 2A 250v
510$

p11-/rod/bnc
10.00Euro

Hanon P810/P850 Automatic Polarimeter
2200$

VISCOMETER VK 2000
600$
Sign up for exclusive sales and product news
© All rights reserved for medical sell

